
Glaucoma is the leading cause of blindness in the world. It is an irreversible chronic disease that affects children and adults; globally, the number of glaucoma patients is expected to increase by 74% by 20401.
In Singapore, nearly 10% of people over the age of 70 have glaucoma2. Considering how glaucoma is largely symptom-free and is hence touted as the “silent thief of sight”, I believe that we still have many unreported cases amongst us.
Unfortunately, many people are still losing vision from glaucoma.
The current mainstay for glaucoma treatment includes eye drops, the success of which highly depends on patient compliance. Furthermore, the patient must adhere to this treatment for life.
If glaucoma eye drops do not lower the eye pressure sufficiently, or if the patient is unable to use the eye drops regularly, conventional glaucoma surgery including trabeculectomy is an option. Trabeculectomy is effective in lowering eye pressure but may be associated with blinding complications including a life-long risk of infection.
Another lesser-known surgical alternative is glaucoma tube implants, which have been used to treat severe glaucoma in the past 30 years with much success. However, until recently, the options for tube implants were limited and the commonly used tube implants were invented more than 20 years ago.
It is time that we take advantage of the advanced technological advancements to develop newer, more effective and safer tube implants for the benefit of our glaucoma patients.
In 2012, my colleague Professor Paul Chew and I, together with a team of clinician-scientists from the National University Health System, started developing a new tube implant – the Paul Glaucoma Implant (PGI). The goal was to create an implant that is better than currently available devices — and we sure did.
Today, the PGI is used in 17 countries worldwide and has greatly improved many patients’ quality of life. Read all about it in this Straits Times feature.

In this article, allow me to explain what glaucoma tube implants are, how the Paul Glaucoma Implant fares against current choices and why I believe this new invention will greatly change the treatment landscape for glaucoma.
With glaucoma, the fluid within the eye does not drain properly. This results in elevated eye pressure (or intraocular pressure) which damages the eye nerve, eventually leading to blindness if untreated.
Hence, the only effective way to treat glaucoma is to reduce eye pressure.
Glaucoma tube implants are devices placed in the eye to allow fluid to drain out of the eye. This helps lower eye pressure and prevents further damage to the eye nerve.
There are a few types of glaucoma implants out there, but regardless of which implant is used, it should be noted that the implants do not improve vision. Instead, they are used to lower eye pressure and prevent further vision loss from glaucoma.
Glaucoma tube implants are typically used in situations such as:
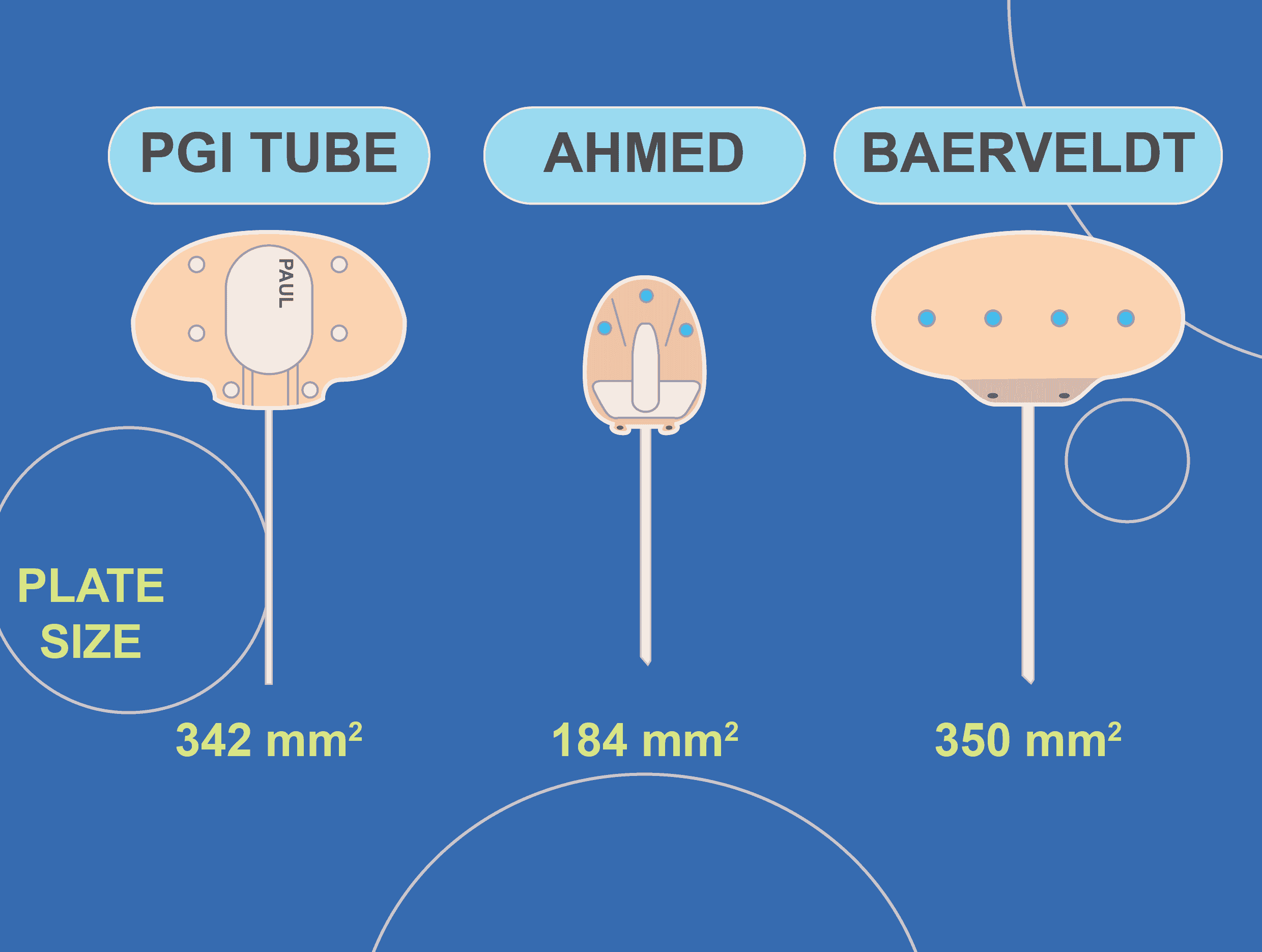
All tube implants have a smaller tub attached to a larger plate.

There are two types of glaucoma implants — valved and non-valved. Respectively, the two most commonly used tube implants are the Ahmed Glaucoma Valve (AGV) and the Baerveldt Glaucoma Implant (BGI). The Baerveldt implant has a larger plate compared with the Ahmed implant (350mm2 vs 184mm2).
The option you choose depends on a few factors, including the severity of your condition and whether you’ve had surgical interventions in the past.
In terms of results and efficacy, comparison studies3 have found that the Baerveldt has a lower failure rate but significantly higher risk4 of hypotony (low eye pressure). The superior efficacy of the Baerveldt implant is thought to be related to its larger plate size. While the valve mechanism of the Ahmed decreases the risk of postoperative complications, it is less effective in lowering the eye pressure compared with a Baerveldt tube4,5.
There is a need for a glaucoma tube implant with similar efficacy to the Baerveldt but with a reduced complication rate, hence the Paul Glaucoma Implant was developed.

Like the Ahmed and Baerveldt tubes, the PGI is an aqueous shunt designed to reduce eye pressure. It can be used for patients with:
The tube size of the PGI (0.1mm) is smaller compared to the Baerveldt and Ahmed tubes (0.3mm). The smaller tube size of the PGI is likely to decrease the risk of complications, including tube exposure, cornea damage and hypotony (low eye pressure).
The plate size of the PGI is similar to the Baerveldt implant and larger than the Ahmed implant, hence it is likely that the efficacy of the PGI is similar to the Baerveldt and better than the Ahmed.

PGI was created with one aim in mind — to design an implant that can achieve better outcomes for our glaucoma patients. After years of tireless research, we can now answer these pertinent questions:
In the clinical trials6 we conducted to study the efficacy of PGI one year after implantation, 93.2% achieved qualified success. 68.9% saw complete success. These results suggest that efficacy of the PGI is similar to the Baerveldt Glaucoma Implant and better than the Ahmed Glaucoma Valve, with a higher safety profile than the Baerveldt Glaucoma Implant.
The surgical technique for implanting the PGI is similar to the Baerveldt implant, with a higher level of complexity compared with the Ahmed Glaucoma Valve. Hence, it is important to find a surgeon who is skilled and trained in implanting the PGI to perform the surgery.

Aqueous flow through the PGI is regulated with an internal suture in the lumen of the tube, so as to prevent the eye pressure from going too low in the early postoperative period. Subsequently, your surgeon will be able to increase the flow through the implant by removing this suture. With meticulous monitoring and management, your surgeon can titrate flow through the PGI after the surgery.
In a study7 conducted at the Manchester Royal Eye Hospital in the UK, 90.1% were qualified successes and achieved an IOP of >5 a year later without the need for medication. I have patients whose eye pressure is still well-controlled (often without glaucoma medications!) a few years after implanting the PGI.
| PGI | Ahmed | Baerveldt | |
| Mean IOP one year post-operation | 13.2±3.3 | 16.57±3.35 | 14.76±2.5 |
| Failure | 5.4% | 19.67% | 12% |
The same study found that the mean number of medications dropped from 3.61 ± 1.09 to 1.22 ± 1.21 post-operation.

As with other implants, potential postoperative complications include;
However, your chances of getting side effects are highly dependent on factors like the severity of the condition and the skill of the surgeon.
Every day, glaucoma is still robbing patients of their vision. While we still need safer and more effective surgical options for glaucoma, with newer devices like PGI developed with intelligent technology and improved surgical outcomes, I believe this puts us a few steps forward and gives us hope of preserving the vision of millions of glaucoma patients worldwide.
If you have any questions regarding the condition or surgery, please feel free to drop me a message and I am more than happy to help.

38 Irrawaddy Road Mt Elizabeth Novena Specialist Centre, #06-25, Singapore 329563
Mon - Fri: 9am - 5pm
Saturday: 9am - 1pm
Sunday / PH: Closed